The versatile excellence on human beings’ part is rooted in many different things, but more importantly than the rest, it is rooted in our ability to grow on a consistent basis. This commitment towards getting …
The versatile excellence on human beings’ part is rooted in many different things, but more importantly than the rest, it is rooted in our ability to grow on a consistent basis. This commitment towards getting better, no matter the circumstances, has enabled the world to clock some huge milestones, with technology emerging as quite a major member of the group. The reason why we hold technology in such a high regard is, by and large, predicated upon its skill-set, which guided us towards a reality that nobody could have ever imagined otherwise. Nevertheless, if we look beyond the surface for one hot second, it will become abundantly clear how the whole runner was also very much inspired from the way we applied those skills across a real world environment. The latter component, in fact, did a lot to give the creation a spectrum-wide presence, and as a result, initiated a full-blown tech revolution. Of course, the next thing this revolution did was to scale up the human experience through some outright unique avenues, but even after achieving a feat so notable, technology will somehow continue to bring forth the right goods. The same has turned more and more evident in recent times, and assuming one new discovery ends up with the desired impact, it will only put that trend on a higher pedestal moving forward.
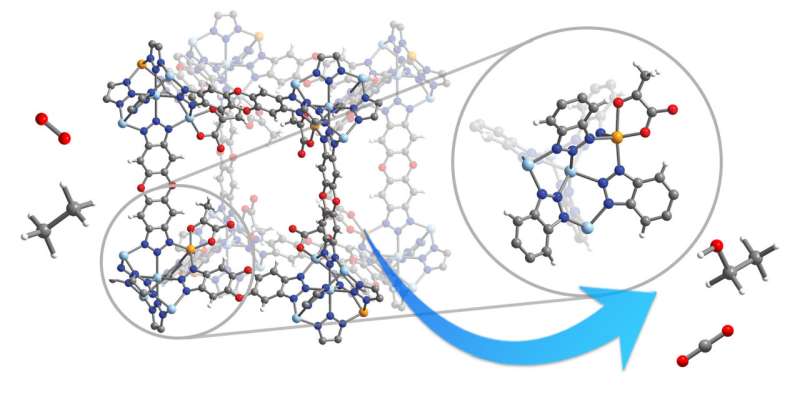
The researching team at University of California Berkeley has successfully developed a method to give a green tech treatment to those natural gases that we burn at oil and gas wells, making up a process called flaring. In order to understand the significance attached to this development, we must dig into why exactly flaring needs the stated treatment. You see, for starters, it is a major waste of fossil fuels, and if that is not big enough problem in your eyes, then we must mention how flaring also poses a massive detriment to our climate. Making the problem even graver is the fact that, according to International Energy Agency, we are currently flaring more than 140 billion cubic meters of natural gas per year. Despite such a big number, capturing this flared gas hasn’t been economically feasible thus far. Enter University of California Berkeley’s latest brainchild. The solution in question focuses on converting primarily methane and ethane into valuable liquids, mostly alcohols like methanol and ethanol. The rationale behind this talks to liquid being an easier commodity to store. Furthermore, the resulting alcohol can be used as feedstocks for production of numerous other petrochemical products, therefore providing an extra bit of revenue source for oil and gas companies. Talk about how the whole process will happen on an actionable note, it involves an oxidization mechanism where an enzyme centered around a reactive metal, in most cases iron, catalyzes the insertion of an oxygen atom between a carbon and hydrogen bond to produce C-O-H, an alcohol group. Now, many studies have already been conducted on finding variants of these natural enzymes that would convert gaseous hydrocarbons from fossil fuels into liquid alcohols without the energy input and huge infrastructure needed today in the chemical industry. However, a bigger chunk of these efforts mandate bringing artificial enzymes in a liquid solution. The new method, on its part, solves the problem by integrating reactive iron sites into a rigid and porous crystalline structure, a metal-organic framework or MOF, which stabilizes the iron and allows easy entry of gas and easy exit of liquid alcohols.
“We have a lot of natural gas wells here that are too small for building a large-scale facility around it to convert ethane to ethanol or methane to methanol,” said Jonas Börgel, a UC Berkeley postdoctoral fellow, who is the author of the paper explaining this study. “This system is nice, in that it is the first purely synthetic and not enzymatic process that can utilize oxygen at near ambient temperature to perform these reactions reminiscent of metalloenzyme reactivity. That’s where it really shows its potential for converting natural gas components into more easily storable energy sources like alcohols. The big advantage of this is converting natural gas into something that has more value than the gas itself.’
The whole effort interestingly sprung to action only after Börgel and its co-author on the study, Kaipeng Hou, now a postdoctoral fellow at UCSF, discovered that an iron-containing variant of the MOF called MFU-4l could be chemically modified to yield an active iron (II) site, which resembles the active site in a natural enzyme called taurine dioxygenase (TauD). TauD, in turn, boasted the means to oxygenate the amino acid taurine.
“We were inspired by the enzyme active site in TauD—the geometry around the iron center looks very similar to the coordination geometry around the peripheral metals in the nodes in this metal organic framework. We thought that if we modified the MOF in the same way and put another ligand on there that is like the cofactor in the enzyme, maybe we would be able to mimic the C-H oxygenation reactivity that the enzyme has. That turned out to be true, said Borgel.
For the purpose of validating their research, the team conducted a detailed spectroscopic analysis of the modified MOF and confirmed that the iron (II) active site was acting just like the iron(II) site in TauD.
Although spearheaded by UC Berkeley, the study saw contributions coming from Argonne National Laboratory, Northwestern University in Illinois, the University of Missouri in Rolla, Berkeley Lab, and UC Davis.